Bile Solubility Test – Principle, Procedure, Results, Quality Control & Limitations
What is Bile Solubility Test?
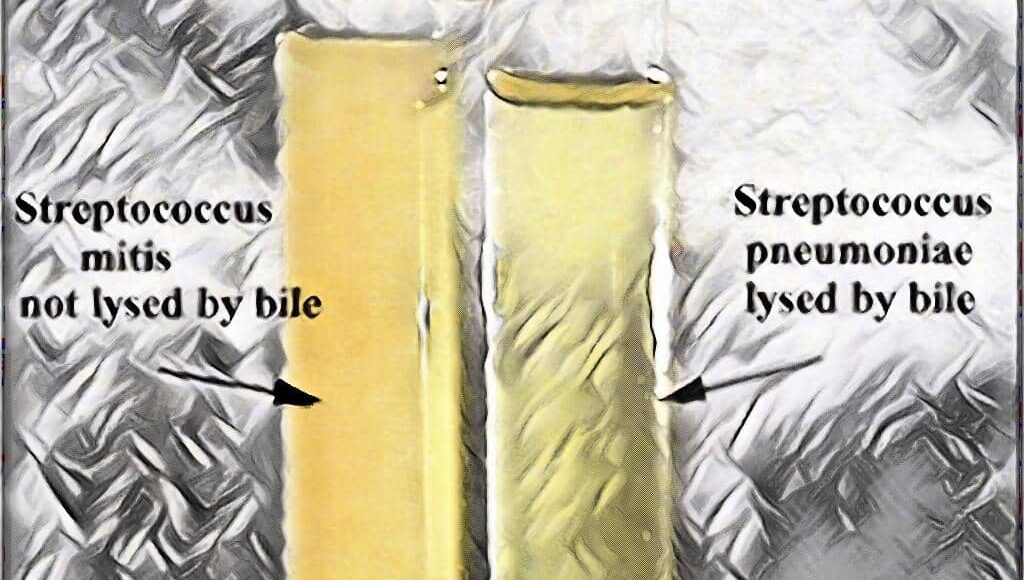
The Bile Solubility Test is one of the most important tests which is commonly used to differentiate the Streptococcus pneumoniae, which is positive for bile solubility test, from the other alpha-hemolytic streptococci like Streptococcusviridans, which is Bile Solubility test Negative.
What is the Principle of Bile Solubility Test?
[wp-svg-icons icon=”point-right” wrap=”i”] The Bile solubility test is primarily used for the identification and differentiation of S. pneumoniae.
[wp-svg-icons icon=”point-right” wrap=”i”] It has an autolytic enzyme which can easily be demonstrated by exposing the bacterial culture to the Bile salts solution.
[wp-svg-icons icon=”point-right” wrap=”i”] In this test, the culture of the test organism is allowed to age in the incubator for at least 24-48 hours or until the desired turbidity of the broth culture attained (See Procedure).
[wp-svg-icons icon=”point-right” wrap=”i”] This culture is now treated with the 2% – 10% Sodium Deoxycholate solution (Bile) under optimum temperature and Time.
[wp-svg-icons icon=”point-right” wrap=”i”] Bile salt solution rapidly lyses the pneumococcal cells present in the solution. This cell lysis depends on the presence of an intracellular autolytic enzyme.
[wp-svg-icons icon=”point-right” wrap=”i”] This is the basis of Bile Solubility test the other Streptococci are Bile Insoluble due to the absence of these Autolytic enzymes.
[wp-svg-icons icon=”point-right” wrap=”i”] Bile salts present in the bile solution acts on the bacterial cell by lowering the surface tension in between the bacterial cell membrane and the surrounding medium which induces the organism’s natural autolytic process. This autolysis causes the clearing of the medium.
[wp-svg-icons icon=”point-right” wrap=”i”] Bile salts lower the surface tension between the bacterial cell membrane and the medium, thus accelerating the organism’s natural autolytic process.
[wp-svg-icons icon=”point-right” wrap=”i”] Bile salts activate the autolytic enzyme and thus the lysis of the bacterial cells which induces clearing of the culture.
[penci_related_posts dis_pview=”yes” dis_pdate=”yes” title=”Editor’s Choice” background=”” border=”” thumbright=”no” number=”4″ style=”grid” align=”none” withids=”” displayby=”cat” orderby=”rand”]Practically, the actual mechanism of Bile Solubility test is not completely understood but the activation of Autolytic enzymes and lysis of bacterial cell is considered as the Basis of this test.
What are the Reagents Required for Bile Solubility Test?
In bile Solubility test mainly two different concentrations of the Bile Salt Solutions are used which are as follows:
2% Bile Salt (Sodium deoxycholate) Solution
[wp-svg-icons icon=”point-right” wrap=”i”] This can easily be prepared in the laboratory, just dissolve 2 grams of sodium deoxycholate powder into 100 ml of Sterile distilled water.
10% Bile Salt (Sodium deoxycholate) Solution
[wp-svg-icons icon=”point-right” wrap=”i”] This can also be prepared in the laboratory just dissolve 10 grams of sodium deoxycholate powder into 100 ml of Sterile distilled water.
Now let’s come to the procedure of Bile Solubility test, here I’m gonna discuss three most commonly employed methods of performing Bile Solubility Test. You can any method as per your convenience…
The procedure of Bile Solubility Test (Tube Method)
[wp-svg-icons icon=”point-right” wrap=”i”] Take 2 ml of Normal Saline Solution (0.85% NaCl) in a clean and dry test tube and prepare a suspension of isolated bacteria in it.
[wp-svg-icons icon=”point-right” wrap=”i”] Now incubate the suspension for at least 24 hours or until it attains the turbidity similar to that of 0.5 – 1 McFarland Standard Solution.
[wp-svg-icons icon=”point-right” wrap=”i”] Divide the Standard Bacterial suspension into two tubes. Adjust the suspension to that of 0.5 – 1 McFarland Standard solution.

[wp-svg-icons icon=”point-right” wrap=”i”] Now take one of the two test tubes, add 2 drops of 2% Bile Salt (Sodium deoxycholate) Solution and mix well the content.
[wp-svg-icons icon=”point-right” wrap=”i”] The other test tube is used as the Control so to this just add 2 drops of Sterile Distilled Water and mix well the content.
[wp-svg-icons icon=”point-right” wrap=”i”] Incubate both the tubes for 10 – 15 minutes at 35 – 37°C.
[wp-svg-icons icon=”point-right” wrap=”i”] Finally, observe the test tube containing 2% Bile Salt Solution for clearing of the turbid solution.
[wp-svg-icons icon=”point-right” wrap=”i”] If turbidity persists, continue to incubate the tubes for up to 3 hours. After that, observe again for clearing.
The procedure of Bile Solubility Test (Plate Method)
[wp-svg-icons icon=”point-right” wrap=”i”] Inoculate the Specimen on 5% Sheep blood agar medium and incubate for 12 – 24 hours at 37°C.
[wp-svg-icons icon=”point-right” wrap=”i”] Now after incubation when the isolated colonies appear on the medium, place one or two drops of 10% Bile Salt Solution to the side of the freshly isolated colony on the Blood agar plate.
[wp-svg-icons icon=”point-right” wrap=”i”] Now gently wash the solution present over the colony using distilled water with dislodging the colony from the blood agar medium plate.
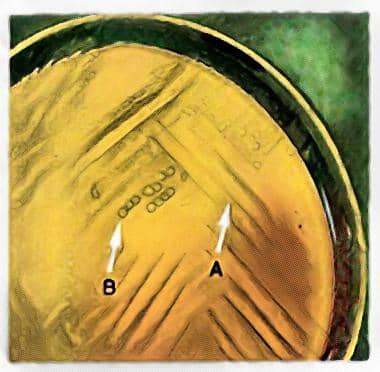
A.) Intact Colony; B.) Lysed Colony
[wp-svg-icons icon=”point-right” wrap=”i”] Incubate the test culture plate at 37°C for about 30 minutes.
[wp-svg-icons icon=”point-right” wrap=”i”] Carefully observe the colony for any lytic activity like the disappearance of the colony.
Common Situation in Bile Solubility Test:
[wp-svg-icons icon=”point-right” wrap=”i”] Sometimes the incomplete clearing (or partially soluble) of the solution observed when the bile salt solutions are added to the medium which cannot be considered as the positive test for Streptococcuspneumoniae identification.
[wp-svg-icons icon=”point-right” wrap=”i”] Those strains of bacteria which are partially soluble in bile solution and have optochin sensitivity zone of inhibition of less than 14 mm are not considered as Streptococcuspneumoniae.
Quality control in Bile Solubility Test
[wp-svg-icons icon=”point-right” wrap=”i”] Always test the new lot of reagents with known positive & negative controls before putting it into Clinical use.
[wp-svg-icons icon=”point-right” wrap=”i”] Discard the Bile Salt solution is not clear, turbid or light amber color.
[wp-svg-icons icon=”point-right” wrap=”i”] Organisms used as a control are as follows:
- Bile Solubility test Positive:
- Streptococcus pneumoniae ATCC 49619
- Streptococcus pneumoniae ATCC 6305
- Bile Solubility Test Negative:
- Enterococcus faecalis ATCC 29212
- Streptococcus sanguis ATCC 10556
Limitations of Bile Solubility Test
[wp-svg-icons icon=”point-right” wrap=”i”] Some Streptococcuspneumoniae strains won’t lyse in the presence of bile salt solution, probably because of the loss of virulence factor or capsule. If lysis isn’t present, the isolate may still be S. pneumonia.
[wp-svg-icons icon=”point-right” wrap=”i”] Therefore, colonies resembling S. pneumoniae that aren’t bile soluble should be further analyzed using another methodology, like optochin sensitivity and/or DNA probe.
[wp-svg-icons icon=”point-right” wrap=”i”] Bile salts won’t induce clearing of a killed culture or one that’s too acid. Thus saline suspensions of young cultures are used.
[wp-svg-icons icon=”point-right” wrap=”i”] The test shouldn’t be performed in old cultures, because the active enzyme may be lost.
[wp-svg-icons icon=”point-right” wrap=”i”] Normal lysis of S. pneumoniae is also inhibited by a high concentration of bile salts being used. Evaporation may cause the reagent to become additionally concentrated, thus affecting the test.
[wp-svg-icons icon=”point-right” wrap=”i”] When performing the bile solubility tube test using saline or unbuffered broth, it is essential to regulate the pH to neutral before adding the reagent in order to avoid false negative reactions.
[wp-svg-icons icon=”point-right” wrap=”i”] Bile solubility is used only to differentiate Streptococcuspneumoniae from other alpha-hemolytic Streptococci.
[wp-svg-icons icon=”point-right” wrap=”i”] When testing using the plate method, care must be taken not to dislodge the colony being tested, therefore resulting in false-positive results. If the direct plate is troublesome to interpret, the test ought to be recurrent using the tube or slide technique.
Frequently Asked Questions (FAQs)
The bile solubility test is used to differentiate Streptococcus pneumoniae from other alpha-hemolytic streptococci. The test result is positive if the bacterial colony is lysed in the presence of bile salts.
Bile is a mixture of bile salts, pigments, and cholesterol. Bile salts can emulsify fats and facilitate their absorption. In the bile solubility test, the bile salts act as a detergent to disrupt the cell membrane of certain bacteria.
The bile solubility test is used to detect the presence of bile salts in a sample. Bile salts are typically tested using the bile-esculin test or the deoxycholate citrate agar test.
The bile solubility test is a microbiological test used to differentiate Streptococcus pneumoniae from other alpha-hemolytic streptococci. S. pneumoniae is bile soluble, while other alpha-hemolytic streptococci are bile resistant.
The bile solubility test is not a confirmatory test on its own, but it can be used in combination with other tests to confirm the identification of S. pneumoniae.
Bile salts are the reagent used in the bile solubility test. A 2% solution of sodium deoxycholate is commonly used for this test.